Edura™ Gentamicin Microspheres: Preventing Surgical Site Infections
Edura™ Gentamicin Microspheres is a first-in-class, therapeutic drug delivery system to prevent surgical site infections. By achieving regulatory approval for clinical testing through FDA and SFDA filings by 2025, sufficient clinical testing could be performed over the subsequent five-year period to have FDA and SFDA approval to market Edura™ Gentamicin Microspheres as a first-in-class therapeutic to prevent surgical site infections by 2030.
Download PDF
Figure 1: Electron micrograph showing Edura™ Gentamicin Microspheres
Value Proposition
Edura™ was developed to address the unmet medical need of surgical site infections. When Edura™ microspheres are added to the surgical wound, the biodegradable microspheres slowly begin to dissolve and release gentamicin into the surrounding tissue spaces, establishing a localized concentration of gentamicin that kills infectious agents such as Pseudomonas aeruginosa and Staphylococcus aureus, two potentially life-threatening bacteria.
Market Potential
The Total Addressable Market (TAM) for surgical site infection-related products and services is estimated at $3.3 billion in the USA. The Serviceable Available Market (SAM) is estimated at $1.1 billion, with a Serviceable Obtainable Market (SOM) of $110 million. If Flow Pharma captures 10% of the SOM, first-year revenues after FDA approval could reach $11 million for the US market alone.
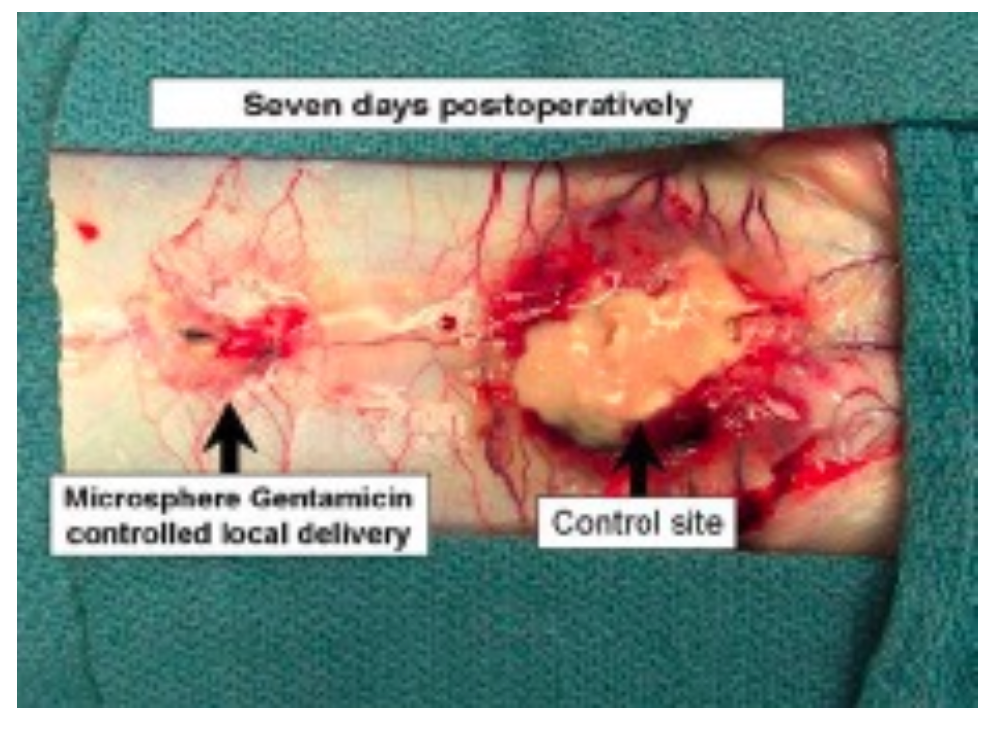
Figure 2: Representative post-mortem rabbit spine image after intra-operative S. aureus challenge of two adjacent spine surgery sites showing significant wound healing differences between a control- and gentamicin-microsphere treated site

